T. Nguyen-Jones1, E. Rogers1, K.K. Wolf2, T. Stone2, S. Presnell1, E.L. LeCluyse2, J. Chen1
1LifeNet Health LifeSciences, Virginia Beach, VA; 2LifeNet Health LifeSciences, Research Triangle Park, NC
Introduction
The rise of chemical production and usage during the 21st century in a wide range of areas such as pharmaceuticals, pesticides, and other industries have come with an increased risk of human exposure to toxic substances which could lead to serious health hazards. Disruption of thyroid hormone synthesis is one of the potential threats that could result in neurodevelopmental impairment and preterm birth. In recent years, different government agencies across the world have increased efforts to develop programs to identify, screen, and determine the potential effects of chemicals found in the environment on thyroid hormone synthesis. These efforts include United States Environmental Protection Agency (US-EPA)’s Endocrine Disruptor Screening Program (EDSP) and the European Chemical Agency (ECHA)’s guidance on endocrine disruptors. Due to the need of screening an extensive list of available chemicals, the demand for a high-throughput approach to replace the conventional low-throughput animal dependent method has become critical. Currently, many of the high-throughput in vitro models used for this purpose often employ microsomes or cell lines which do not always accurately represent the human thyroid physiological conditions or native properties especially the follicular structure that is responsible for thyroid hormone synthesis. Therefore, the development of more physiologically relevant human models has become an emergent need. The goal of this study was to evaluate the use of P1 cryopreserved human thyrocytes from healthy donors in the 3D thyroid microtissue assay for determining the response profiles of different thyroid-disrupting chemicals (TDCs).
Materials & Methods
Cryopreserved thyrocyte cell cultures
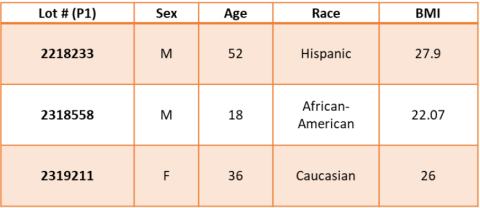
Thyrocytes from healthy male and female donors (18-52 years of age, n = 3) were isolated using previously described procedure1. The cryopreserved thyrocytes were thawed (p1)and plated in 3D cell culture format using Matrigel-coated 96-well plates. On day 2 of cultures, cells were stimulated with 1.0 mIU/mL bovine thyroid stimulating hormone (bTSH) allowing microtissue formation until day 8. On day 8, cell cultures were dosed with either known TPO inhibitor (methimazole), or NIS inhibitor (potassium hexafluorophosphate KPF6) at 100 – 0.0001 μM, or anti-TSHR (thyroid-stimulating hormone receptor) recombinant human antibody K1-70 at 0.667 – 6.67x10-7 μM.
Assay analysis
On day 14, media samples from cell cultures were collected for T4 level quantification using InvitrogenTM Thyrocyte T4 Competitive ELISA kit (EIAT4C). Cells’ viability were determined using Promega CellTiter-Glo® Luminescent Cell Viability Assay (G7570).
For histology, microtissues were fixed using formalin and proceeded with conventional processing and H&E staining procedure.
Data analysis
Data analysis was performed using Microsoft Excel and IC50 values were determined using GraphPad Prism software.
Results
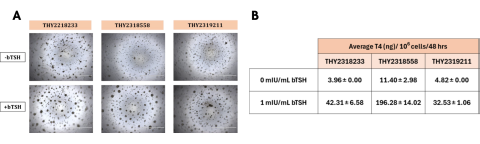

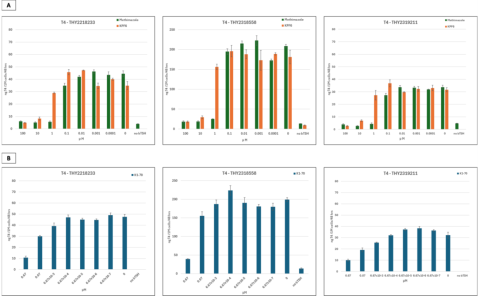
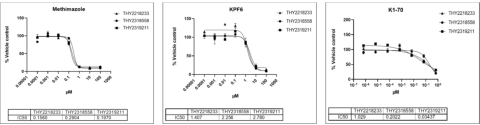

Conclusions
- Follicular structures of the native thyroid were successfully reconstructed for T4 synthesis via culturing of cryopreserved thyrocytes (P1) in a 3D cell culture format using Matrigel-coated 96-well plates.
- The response profiles of thyroid hormone T4 to both small molecule and antibody-based reference compounds were achieved in a dose response manner.
- 3D thyrocyte microtissue model offers a platform for environmental chemical screening as well as a potential promising approach for thyroid antibody-based therapeutic research.
References/Acknowledgements
We would like to thank Davorka Softic for her contribution to the H&E microtissue staining work.
Reference:
1. Deisenroth, C., Soldatow, V. Y., et. al. (2020). Development of an in vitro human thyroid microtissue model for chemical screening. Toxicol Sci, 174(1), 63-78. doi:10.1093/toxsci/kfz238.
